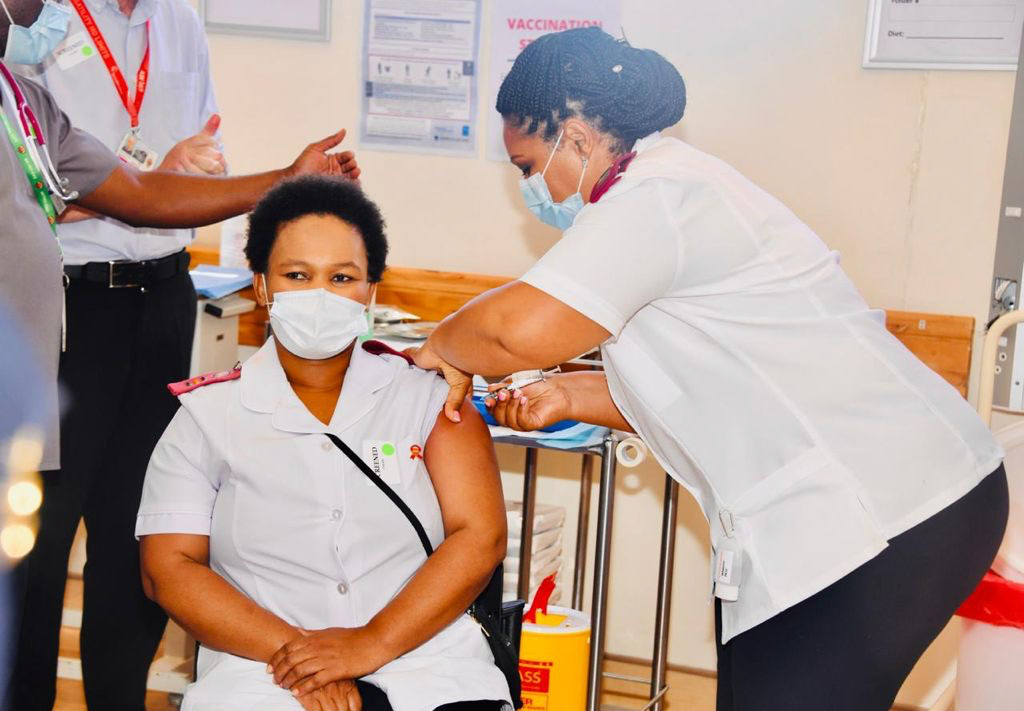
South Africa commences early access vaccine rollout to Healthcare Workers - Sisonke! Let’s work together to protect our Healthcare Workers
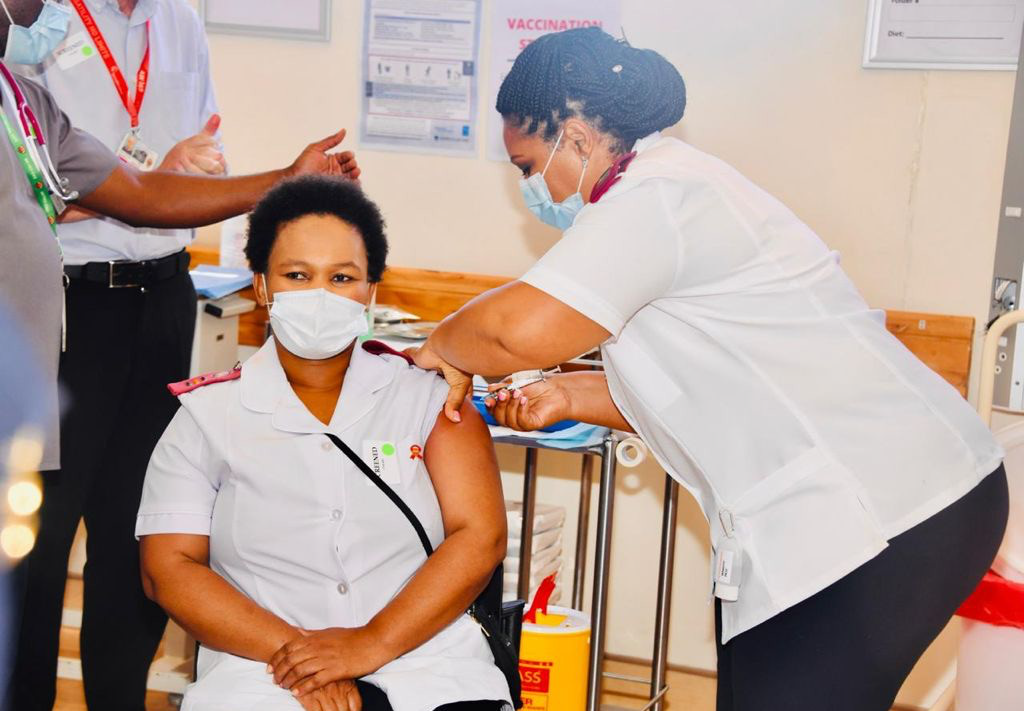 Day One of the first phase of COVID-19 Vaccine Programme to health workers was a huge success today. South Africa secured the single dose Johnson & Johnson (JnJ) vaccine for use in the Sisonke (‘Together’) programme. The Sisonke Study aims to reach up to 500,000 healthcare workers starting in 18 public sector hospitals across all nine provinces.
Day One of the first phase of COVID-19 Vaccine Programme to health workers was a huge success today. South Africa secured the single dose Johnson & Johnson (JnJ) vaccine for use in the Sisonke (‘Together’) programme. The Sisonke Study aims to reach up to 500,000 healthcare workers starting in 18 public sector hospitals across all nine provinces.
As a demonstration of confidence in the safety and efficacy of the vaccine … of organised labour leadership will be taking their shots at Steve Biko Academic Hospital from 14:00. These are leaders who are health workers themselves. They are from: DENOSA, HOSPERSA, NEHAWU, NAPSU, NUPSAW, YNITU, PSA, PHSDSBC, SAEPU, SAMATU.
Protecting our healthcare workers is an urgent priority. Phase 1 of the vaccine programme is focused on healthcare workers. This is the first step in rolling out a comprehensive vaccine distribution strategy for the nation. Healthcare workers are the frontline of the country’s COVID-19 response and are 3-4 times more likely to contract COVID- 19 than the general population. To date 40,000 South African health workers have contracted COVID-19, 6,473 have been hospitalised and 663 of our colleagues have passed on.
Phase 1 of South Africa’s three phase vaccine rollout strategy was paused temporarily earlier this month following concerns regarding the efficacy of the Oxford-AstraZeneca (also known as CoviShield) vaccine against mild-moderate COVID caused by the 501.V2 variant. CoviShield may still play a role in the National Programme, however more research is needed to determine how and when it will be used. Vaccination using a safe and efficacious vaccine must start without delay to protect healthcare workers during a likely 3rd wave this winter. The switch to the JnJ vaccine allows for the country to restart Phase 1 vaccine rollout to keep our health workers safe.
The single dose JnJ vaccine protects against severe COVID-19 in South Africa. The safety and efficacy of the vaccine was assessed in the international Phase 3 study, ENSEMBLE, which was conducted across Latin America, USA and South Africa with more than 43 000 participants. South Africa contributed around 7,000 participants to the trial between October 2020 and February 2021, including participants who were exposed to the 501.V2 variant. The long-term follow-up of study participants is still ongoing, as is required for the process of regulatory approval by the South African Health Products Regulatory Authority (SAHPRA) for commercial or emergency use. However, since the safety and efficacy of the vaccine has already been proven, the current rollout is being conducted under The Sisonke Open Label Program, to assess real world effectiveness among health care workers, who have already been prioritised for vaccination by the South African Government’s Covid-19 Vaccination Strategy.
The JnJ vaccine has been shown to be safe, with no safety concerns reported from past trials or rollouts. The ENSEMBLE trial showed excellent protection of a single-dose vaccine against severe COVID-19, including in South Africa, and as such a delay in rolling out the vaccine would be unethical.
There is a clear process for vaccine rollout to healthcare workers. To receive the vaccine, healthcare workers must: (1) register on the South Africa Electronic Vaccination Data System, (2) respond to an SMS invitation, and (3) provide informed consent to take part. Healthcareworkers will receive a vaccination voucher and communication once a vaccination centre isopentorendertheservice.Toregister,followthislink:https://vaccine.enroll.health.gov.za/#/
The Sisonke rollout will include meaningful collaboration between the original team who were involved in the ENSEMBLE trial, JnJ, and the NDoH and the SAMRC. The partners will closely monitor, track and assess the real-world rollout of the vaccine. They will measure: (1) the occurrence of hospitalizations and deaths, (2) the incidence of severe SARS CoV-2 infections, (3) diversity of breakthrough infections; and (4) evaluate vaccine uptake among healthcare workers in South Africa.
This experienced team will utilise their trained and qualified staff including pharmacists, clinicians and others to receive, store and oversee the dispensation of the JnJ vaccine. Sisonke staff will work very closely with national and provincial health public and private vaccine centres to ensure that the vaccination of healthcare workers is done safely and carefully managed.
The protection of our healthcare workers through the rollout of the safe and efficacious vaccine is an urgent priority for South Africa. As a demonstration
- Sisonke Vaccine Programme - Protecting Healthcare Workers - Fact Sheet
- South Africa commences early access vaccine rollout to Healthcare Workers - Sisonke! Let’s work together to protect our Healthcare Workers - Department of Health - Media Statement (17 February 2021)
Sisonke Study - Protecting Healthcare Workers (Website)
MEDIA ENQUIRIES:
Mr. Popo Maja Spokesperson
National Department of Health
Cell: 072 585 3219/082 373 1169
E-mail: Popo.maja@health.gov.za
Issued by: Department of Health
Date: Wednesday, 17 February 2021